For ten people struck by COVID-19, one dose of antibodies taken from the blood of people who had recovered from the virus appeared to save lives, cut down the duration of the symptoms, raise oxygen levels and quicken viral clearance, according to recent research.
The good news emerged from a “pilot study” published recently in the Proceedings of the National Academies of Sciences (PNAS) journal. The study was conducted in three Chinese hospitals and showed promise of extracting immune antibodies from recovered patients (the process is also referred to as convalescent plasma) and administering them to those fighting with a serious case of COVID-19.
The therapy could potentially be a strong antidote for COVID-19 patients struggling with shortness of breath.
Interestingly, in the early part of the 20th-century doctors transferred the blood-based antibodies of patients who had recovered from mumps, flu, and polio to people who were still suffering from those infections. Thanks to a veteran infectee’s immune system memory of the virus, people receiving convalescent plasma seemed to recover much quicker and in more complete termes than those who did not receive the treatment, it was observed.
And with a proper anti-coronavirus vaccine at least one year away and no additional treatments currently on the market, the United States Food and Drug Administration last month approved the use of such therapy as an experimental treatment in clinical trials and for people in critical condition without any other options available.
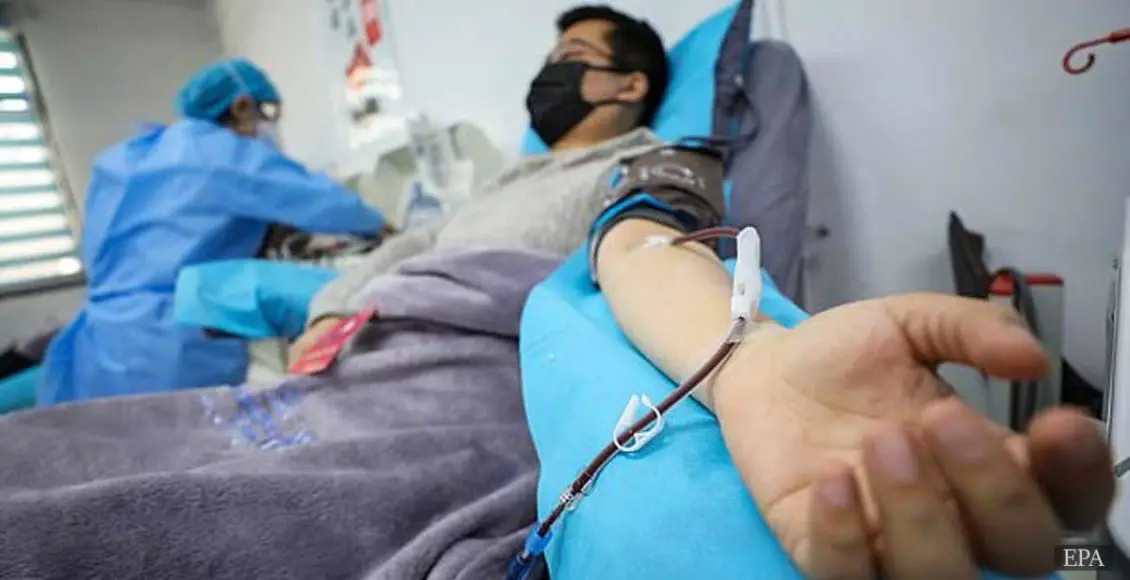
How does it work?
Blood banks receive plasma donations just like they accept donations of pure blood. If a person has decided to only donate plasma, their blood is drawn through a tube, the plasma is separated and the rest is injected back into the donor’s body.
Afterward, the plasma is being tested and purified to make sure it does not contain any viruses and is safe to use.
Currently, Japanese pharmaceutical company Takeda is developing a drug that contains recovered patients antibodies in a pill form, according to Stat News.
Julie Kim, the president of Takeda’s blood plasma unit, said:
“We are not looking at this as a therapy that everyone should go on.
“This will be targeted to patients who have severe disease.”
Kim was not able to comment on exact timelines because Takeda has not yet had discussed the matter with regulators like the Food and Drug Administration.
What are your thoughts on this potential method of recovery? Let us know by joining the conversation in the comments and please share this article to spread the positive news.