China tried to patent coronavirus drug only a day after confirming COVID-19 was human-transmissible
In China, a drug believed to be one of the most effective potential weapons against coronavirus was filed for a patent, the day after they confirmed human transmission of the disease.
This revelation may indicate that China’s perception of the deadly virus may be far advanced from what they claim it to be. Moreover, as reported by Mail Online, it raises severe concerns about a cover-up of the pandemic when it first appeared in Wuhan.
On Friday, April 24th, the chairman of the Foreign Affairs Select Committee, Tom Tugendhat, joined the growing global clamor for a full, independent inquiry into China’s role. He stated:
“It is quite clear there is an awful lot that we don’t know about the emergence of this disease and the responses to it. We all need to learn the lessons of the outbreak so the international community can respond better in the future.”
Presently, the leaders of China’s Communist Party are being accused of covering up data about the actual statistics of coronavirus cases in the country. They are being under the suspicion of blocking public health teams from investigating and silencing healthcare workers of warning the world about the epidemic.
President Xi Jinping confirmed the contagious nature of COVID-19 on January 20th. However, leaked data has shown that there was a serious delay in admitting the virus includes human transmission.
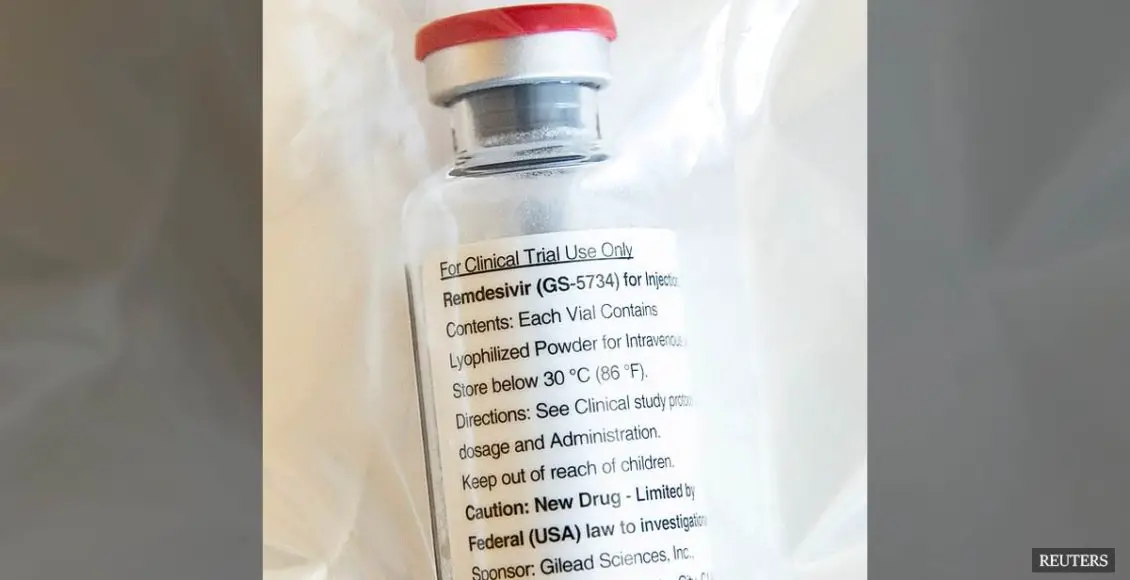
Only a day after, on January 21st, China filed a patent for commercial use of Remdesivir, a drug initially created to fight ebola.
What’s more, the patent application was made by the top-secret bio-laboratory at the Wuhan Institute of Virology, the center of concerns about a possible leak of coronavirus.
Remdesivir was created and developed by Gilead Sciences, a California based biopharmaceutical company that researches, develops, and commercializes drugs.
They claim they have filed their own global applications for Remdesivir’s use against coronavirus four years ago. Currently, countries, companies, and scientists from all over the globe are searching for effective treatments and vaccines against the virus. Those who find the cure for COVID-19 will gain immense prestige.
The California biopharmaceutical company assures that they are aware of what the Chinese have done, but they had no influence over the patent office’s decision. Besides, Gilead cannot comment, since precise details of the application would not be published earlier than next year. They add:
“Our focus at this time is on rapidly determining the potential for Remdesivir as a treatment for Covid-19 and accelerating manufacturing in anticipation of potential future supply needs.”
Even President Donald Trump hailed the drug as ‘promising’ after informal showed it had helped some COVID-19 patients.
Allegedly, it is already being given to certain coronavirus cases on compassionate grounds.
However, Remdesivir still could not be included in the world’s biggest trial of possible treatments being run by Oxford University. Last week, after the results of an initial trial in China that raised concerns leaked, a leading US epidemiologist said it offered ‘hope’ but ‘won’t likely be a home-run drug’.
On the contrary, Gilead argued this study was halted because of low patient enrolment. They claim that the results of a major US government trial would be released in May. This will show whether the drug will improve outcomes such as survival rates in coronavirus patients.
The leader of the Oxford study, Professor Martin Landray, suggests medics will probably end up with a range of medicaments against the deadly virus. He says these drugs might be used in combinations to help reduce fatality rates. Prof. Landray expressed his position:
“It is unlikely we will get a wonder drug that will knock out the infection. Even if you find a drug that reduces the death rate by one fifth, that would have meant we would have been able to save about 4,000 lives already in Britain.”


